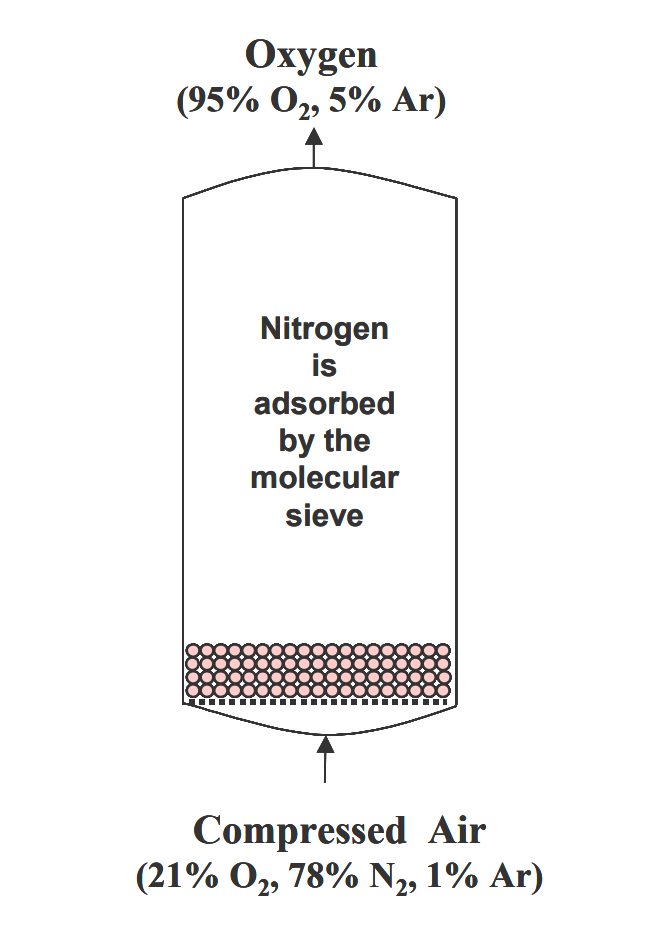
Air contains 21% oxygen, 78% nitrogen, 0.9% argon, and 0.1% other gases.
Both nitrogen and oxygen have quadrupole moments, but the quadrupole moment of nitrogen (0.31A) is much larger than that of oxygen (0.10A), so the adsorption capacity of nitrogen on the zeolite molecular sieve is stronger than that of oxygen (nitrogen and molecular sieve surface ions are strong, as shown in the figure). Therefore, when the air under pressure through the adsorption bed with zeolite molecular sieve adsorbent, nitrogen is adsorbed by the molecular sieve, oxygen is less adsorbed, enriched in the gas phase and out of the adsorption bed, so that oxygen and nitrogen are separated to obtain oxygen.
When the molecular sieve adsorption of nitrogen gas to close to saturation, stop the air and reduce the pressure of the adsorption bed, the molecular sieve adsorption of nitrogen gas can be desorbed, the molecular sieve is regenerated and reused. Two or more adsorption beds can be switched in turn to produce oxygen continuously.
The boiling points of argon and oxygen are close to each other, so it is difficult to separate them and they are enriched together in the gas phase. Therefore, the oxygen concentration produced by the PSA unit is generally less than 95% (the limit concentration of oxygen is 95.6%, and the rest is argon), but the oxygen concentration designed for the unit can be varied in a wide range (60%-95%) according to the user’s requirements, which is very flexible.